


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 14-10-2018
Date: 4-11-2018
Date: 15-10-2018
|
The ammonia acts as both a base and a ligand. With a small amount of ammonia, hydrogen ions are pulled off the hexaaqua ion exactly as in the hydroxide ion case to give the same neutral complex.
That precipitate dissolves if you add an excess of ammonia. The ammonia replaces water as a ligand to give hexaamminecobalt(II) ions.
The color changes are:
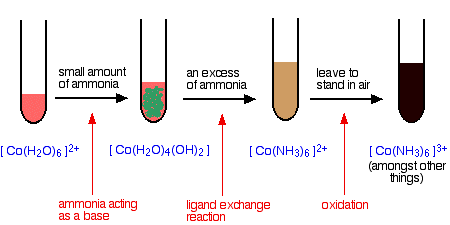
The hexaamminecobalt(II) complex is very easily oxidized to the corresponding cobalt(III) complex. In the test-tube this is seen as a rapid darkening to a deep red-brown solution. In fact the hexaamminecobalt(III) ion is yellow! What you see is a mixture of this ion and various other cobalt(III) ions involving ligand exchange reactions with both water molecules and negative ions present in the solution.



|
|
|
|
10 دقائق قبل النوم.. هذا الأسلوب يساهم في تحسين يومك
|
|
|
|
|
|
|
"جيتكس غلوبال" يرسم ملامح مستقبل التكنولوجيا عالميا
|
|
|
|
|
|
|
شعبة التوجيه الديني النسوي تواصل تقديم الدورة التطويرية لتعليم مناسك الحج
|
|
|