


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 28-10-2020
Date: 25-12-2015
Date: 2-5-2016
|
The lac Repressor Is Controlled by a Small-Molecule Inducer
KEY CONCEPTS
- An inducer functions by converting the repressor protein into a form with lower operator affinity.
- The lac repressor has two binding sites, one for the operator DNA and another for the inducer.
- The lac repressor is inactivated by an allosteric interaction in which binding of the inducer at its site changes the properties of the DNA-binding site.
- The true inducer is allolactose, not the actual substrate of β-galactosidase.
The ability to act as an inducer or a corepressor is highly specific. Only the substrate/product of the regulated enzymes or a closely related molecule can serve this function. In most cases, though, the activity of the small molecule does not depend on its interaction with the target enzyme. For the lac system the natural inducer is not lactose, but rather a by-product of the LacZ enzyme, allolactose. Allolactose is also a substrate of the LacZ enzyme, so it does not persist in the cell. Some inducers resemble the natural inducers of the lac operon but cannot be metabolized by the enzyme. The best example of this is isopropylthiogalactoside (IPTG), one of several thiogalactosides with this property. IPTG is not metabolized by β-galactosidase; even so, it is a very efficient inducer of the lac genes.
Molecules that induce enzyme synthesis but are not metabolized are called gratuitous inducers. The existence of gratuitous inducers reveals an important point. The system must possess some component, distinct from the target enzyme, that recognizes the appropriate substrate, and its ability to recognize related potential substrates is different from that of the enzyme. The separate component that represses the lac operon is the lac repressor protein, which is encoded by the lacI gene. The lac repressor protein is induced by allolactose and IPTG to allow expression of lacZYA. The LacZ enzyme (β-galactosidase) utilizes allolactose and lactose as substrates. lacI is not induced by lactose, and the LacZ enzyme does not metabolize IPTG.
The component that responds to the inducer is the repressor protein encoded by lacI. Its target, the lacZYA structural genes, is transcribed into a single mRNA from the promoter just upstream of lacZ. The state of the repressor determines whether this promoter is turned off or on:
- FIGURE 1shows that in the absence of an inducer the genes are not transcribed, because the repressor protein is in an active form that is bound to the operator.
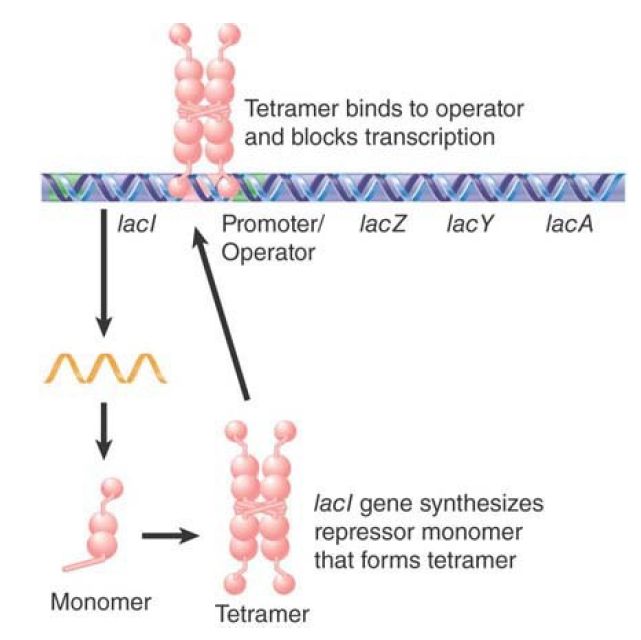
FIGURE 1. The lac repressor maintains the lac operon in the inactive condition by binding to the operator. The shape of the repressor is represented as a series of connected domains as revealed by its crystal structure.
FIGURE 2. shows that when an inducer is added, the repressor is converted into either a form with lower affinity for the operator or a lower affinity form that leaves the operator. Transcription then starts at the promoter and proceeds through the genes to a terminator located beyond the 3′ end of lacA.

FIGURE 2. Addition of the inducer converts the repressor to a form with low affinity for the operator. This allows RNA polymerase to initiate transcription.
The crucial features of the control circuit reside in the dual properties of the repressor: It can prevent transcription, and it can recognize the small-molecule inducer. The repressor has two types of binding site: one type for the operator DNA and one type for the inducer. When the inducer binds at its site, it changes the structure of the protein in such a way as to influence the activity of the operator-binding site. The ability of one site in the protein to control the activity of another is called allosteric control.
Induction accomplishes a coordinate regulation: All the genes are expressed (or not expressed) in unison. The mRNA is translated sequentially from its 5′ end, which explains why induction always causes the appearance of β-galactosidase, β-galactoside permease, and β-galactoside transacetylase, in that order. Translation of a common mRNA explains why the relative amounts of the three enzymes always remain the same under varying conditions of induction. Usually, the most important enzyme is first in the operon.
The constitution of the lac operon has several potential paradoxes. First, the lac operon contains the structural gene (lacZ) coding for the β-galactosidase activity needed to metabolize the sugar; it also includes the gene (lacY) that codes for the protein needed to transport the substrate into the cell. If the operon is in a repressed state, how does the inducer enter the cell to start the process of induction? The second paradox is that β-galactosidase (encoded by lacZ) is required to make the inducer allolactose to induce the synthesis of β-galactosidase. How is allolactose synthesized to allow induction of the gene? (An operon with a mutant lacZ gene cannot be induced.)
Two features ensure induction of the lac operon. First, the operon has a basal level of expression, ensuring that a minimal amount of LacZ and LacY proteins are present in the cell—enough to start the process. Even when the lac operon is not induced, it is expressed at a residual level (0.1% of the induced level). In addition, some inducer enters the cell via another uptake system. The basal level of β-galactosidase then converts some lactose to allolactose, leading to induction of the lac operon.

|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|