


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 28-8-2018
Date: 28-8-2018
Date: 16-1-2022
|
Radical Reactions
Radical reactions are not as common as polar reactions but are nevertheless important in some industrial processes and biological pathways. Let’s see briefly how they occur. A radical is highly reactive because it contains an atom with an odd number of electrons (usually seven) in its valence shell, rather than a stable, noblegas octet. A radical can achieve a valence-shell octet in several ways. For example, the radical might abstract an atom and one bonding electron from another reactant, leaving behind a new radical. The net result is a radical substitution reaction.

Alternatively, a reactant radical might add to a double bond, taking one electron from the double bond and yielding a new radical. The net result is a radical addition reaction.

An example of an industrially useful radical reaction is the chlorination of methane to yield chloromethane. This substitution reaction is the first step in the preparation of the solvents dichloromethane (CH2Cl2) and chloroform (CHCl3).

Like many radical reactions in the laboratory, methane chlorination requires three kinds of steps: initiation, propagation, and termination.
Initiation Irradiation with ultraviolet light begins the reaction by breaking the relatively weak Cl-Cl bond of a small number of Cl2 molecules to give a few reactive chlorine radicals.

Propagation Once produced, a reactive chlorine radical collides with a methane molecule in a propagation step, abstracting a hydrogen atom to give HCl and a methyl radical (-CH3). This methyl radical reacts further with Cl2 in a second propagation step to give the product chloromethane plus a new chlorine radical, which cycles back and repeats the first propagation step. Thus, once the sequence has been initiated, it becomes a selfsustaining cycle of repeating steps (a) and (b), making the overall process a chain reaction.

Termination Occasionally, two radicals might collide and combine to form a stable product. When that happens, the reaction cycle is broken and the chain is ended. Such termination steps occur infrequently, however, because the concentration of radicals in the reaction at any given moment is very small. Thus, the likelihood that two radicals will collide is also small.

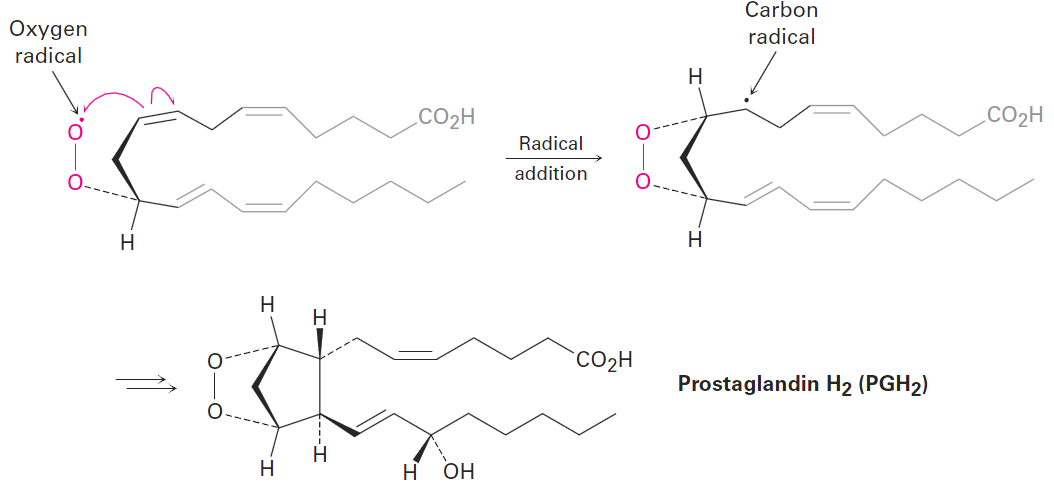
As a biological example of a radical reaction, look at the synthesis of prostaglandins, a large class of molecules found in virtually all body tissues and fluids. A number of pharmaceuticals are based on or derived from prostaglandins, including medicines that induce labor during childbirth, reduce intraocular pressure in glaucoma, control bronchial asthma, and help treat congenital heart defects.
Prostaglandin biosynthesis is initiated by the abstraction of a hydrogen atom from arachidonic acid by an iron–oxygen radical, thereby generating a new, carbon radical in a substitution reaction. Don’t be intimidated by the size of the molecules; focus on the changes occurring in each step. (To help you do that, the unchanged part of the molecule is “ghosted,” with only the reactive part clearly visible.)

Following the initial abstraction of a hydrogen atom, the carbon radical then reacts with O2 to give an oxygen radical, which reacts with a C=C bond within the same molecule in an addition reaction. Several further transformations ultimately yield prostaglandin H2.



|
|
|
|
دراسة يابانية لتقليل مخاطر أمراض المواليد منخفضي الوزن
|
|
|
|
|
|
|
اكتشاف أكبر مرجان في العالم قبالة سواحل جزر سليمان
|
|
|
|
|
|
|
اتحاد كليات الطب الملكية البريطانية يشيد بالمستوى العلمي لطلبة جامعة العميد وبيئتها التعليمية
|
|
|