


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 17-4-2021
Date: 20-5-2021
Date: 1-11-2020
|
Avidin
Avidin is a minor glycoprotein component of egg white that binds the vitamin biotin with the largest association constant known (Ka ~ 1015 M–1) for a ligand–protein interaction (1). Its function in egg white is not known precisely, although protective or antibiotic activities have been suggested. It gained importance not because of its natural functionality, but because of its utility as a tool in molecular biology. Together with its bacterial relative streptavidin and in complex with biotin, avidin has become one of the most important tools in molecular biology because of its myriad of applications in many fields of biology, biotechnology, and even chemistry (2) . Originally, the avidin-biotin complex was introduced to localize, detect, and isolate biologically active molecules (3, 4), but over the years it has became a standard system for replacing radioactivity in immunoassays, for DNA probes, and for various clinical and medical applications (5, 6).
The avidin molecule forms a very stable tetramer that consists of four identical monomers. The monomer is glycosylated at asparagine-17 and is strongly basic (isoelectric point of about 10.5) due to a surplus of arginine and lysine residues. The relatively high molar absorption (A1% [280 nm] = 1.54) reflects the four tryptophan and one tyrosine residue per monomer. The molecular mass of the glycosylated tetramer is about 62,400 Da. When the oligosaccharide residue is removed, the estimated mass is 57,120 Da.
Each avidin monomer binds one molecule of biotin. The binding is considered to be kinetically noncooperative among the subunits (7), although one of the tryptophan residues from a neighboring monomer is inserted physically into the binding site and thus contributes to binding biotin and to the enhanced stability of the tetrameric structure (8). Avidin also binds to the dye HABA (4′-hydroxyazobenzene-2-carboxylic acid), but the association constant (ca. 106 M–1) is considerably lower than that of the avidin-biotin complex.
The unusually strong binding and the many uses of the system caused scientists to try to understand the molecular basis for this interaction. Moreover, since its inception, many groups have sought to improve various components of the system or the system as a whole. Concurrently, structure-function studies have been implemented, in which early results show the importance of aromatic amino acids in the biotin-binding sites of both avidin and streptavidin. The final structural proof is the determination of the crystal structure of both proteins (Fig. 1), which shows that their overall fold, organization into the tetramer, and content and position of amino acids in the binding sites are all similar (8-10).
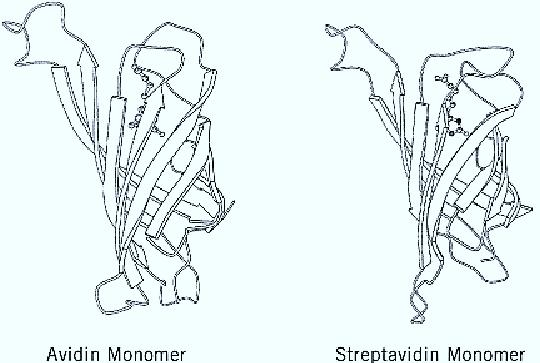
Figure 1. Ribbon diagrams of the avidin–and streptavidin–biotin monomers, showing the eight strands of the b-barrel. The biotin molecule is shown in a ball-and-stick model.
The avidin monomer forms a b-barrel, composed of eight antiparallel b-strands, connected sequentially by loops. The biotin-binding site is a hydrophobic pocket that, in the absence of biotin, contains five molecules of structured water. The combined structure of these water molecules emulates that of biotin itself, and as a consequence, they prevent the binding pocket from collapsing. The water molecules are expelled upon binding of biotin. Because no conformational change occurs during complex formation, the binding has a gain of free energy. In fact, the avidin-biotin system can be considered a natural host–guest complex. It is clear that any change of this “ideal system” leads to reduced affinity. Indeed, site-directed mutagenesis or chemical modification of residues in
the binding site have reduced the affinity constant. An example is the nitration of the essential binding-site tyrosine (11), which decreases the association constant to ~1010 M–1 , and renders the binding reversible at alkaline pH. Another example is the site-directed modification of tryptophan 120 in streptavidin to phenylalanine, which is also accompanied by a loss in affinity (12).
Different modifications of avidin were introduced over the years, including Extravidin, Neutralite avidin, and Lite avidin (13). Extravidin and Neutralite avidin are avidins in which the arginine residues were modified, whereas the oligosaccharide moieties were removed from Lite and Neutralite avidins. These derivatives are useful in many applications, because avidin, being a basic glycoprotein, is subject to a variety of extraneous nonspecific or undesired binding, particularly to DNA. Similar qualities of genetically engineered forms of avidin are possible because avidin was successfully expressed in a eukaryotic host (14, 15). This should enable preparation of different derivatives of avidin with high affinities, provided that the binding site residues are preserved. It seems that the nonbinding-site residues can be altered extensively .
References
1. N. M. Green (1975) Adv. Protein Chem. 29, 85–133.
2. M. Wilchek and E. A. Bayer (1989) Trends Biochem. Sci. 14, 408–412.
3. E. A. Bayer and M. Wilchek (1978) Trends Biochem. Sci. 3, N237–N239.
4. E. A. Bayer and M. Wilchek (1980) Methods Biochem. Anal. 26, 1–45.
5. M. Wilchek and E. A. Bayer (1984) Immunol. Today 5, 39–43.
6. M. Wilchek and E. A. Bayer (1988) Anal. Biochem. 171, 1–32.
7. M. L. Jones and G. P. Kurzban (1995) Biochemistry 34, 11750–11756.
8. O. Livnah, E. A. Bayer, M. Wilchek, and J. L. Sussman (1993) Proc. Natl. Acad. Sci. USA 90, 5076–5080
9.W. A. Hendrickson, A. Pähler, J. L. Smith, Y. Satow, E. A. Merritt, and R. P. Phizackerley (1989) Proc. Natl. Acad. Sci. USA 86, 2190–2194.
10. P. C. Weber, D. H. Ohlendorf, J. L. Wendoloski, and F. R. Salemme (1989) Science 243, 8588 –
11. E. Morag, E. A. Bayer, and M. Wilchek (1996) Biochem. J. 316, 193–199.
12.T. Sano and C. R. Cantor (1995) Proc. Natl. Acad. Sci. USA 92, 3180–3184.
13. E. A. Bayer and M. Wilchek (1994) In Egg Uses and Processing Technologies (J. S. Sim, and S. Nakai, eds.), CAB International, Wallingford, UK, pp. 158–176.
14. K. J. Airenne, P. Sarkkinen, E.-L. Punnonen, and M. S. Kulomaa (1994) Gene 144, 75–80.
15. K. J. Airenne, C. Oker-Blom, V. S. Marjomäki, E. A. Bayer, M. Wilchek, and M. S. Kulomaa (1997) Protein Express. Purif. 9, 100–108.

|
|
|
|
مخاطر عدم علاج ارتفاع ضغط الدم
|
|
|
|
|
|
|
اختراق جديد في علاج سرطان البروستات العدواني
|
|
|
|
|
|
|
مدرسة دار العلم.. صرح علميّ متميز في كربلاء لنشر علوم أهل البيت (عليهم السلام)
|
|
|