


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية| Pre-mRNA Splicing Likely Shares the Mechanism with Group II Autocatalytic Introns |
|
|
|
Read More
Date: 29-12-2015
Date: 30-11-2015
Date: 2-4-2021
|
Pre-mRNA Splicing Likely Shares the Mechanism with Group II Autocatalytic Introns
KEY CONCEPTS
- Group II introns excise themselves from RNA by an autocatalytic splicing event.
- The splice sites and mechanism of splicing of group II introns are similar to splicing of nuclear introns.
- A group II intron folds into a secondary structure that generates a catalytic site resembling the structure of a U6–U2 nuclear intron.
Introns in all genes (except nuclear tRNA–encoding genes) can be divided into three general classes. Nuclear pre-mRNA introns are identified only by the presence of the GU … AG dinucleotides at the 5′ and 3′ ends and the branch site/pyrimidine tract near the 3′ end. They do not show any common features of secondary structure. In contrast, group I and group II introns found in organelles and in bacteria (group I introns are also found in the nucleus in unicellular/oligocellular eukaryotes) are classified according to their internal organization. Each can be folded into a typical type of secondary structure.
The group I and group II introns have the remarkable ability to excise themselves from an RNA. This is called autosplicing, or self-splicing. Group I introns are more common than group II introns. There is little relationship between the two classes, but in each case the RNA can perform the splicing reaction in vitro by itself, without requiring enzymatic activities provided by proteins; however, proteins are almost certainly required in vivo to assist with folding .
FIGURE 1 shows that three classes of introns are excised by two successive transesterifications (shown previously for nuclear introns). In the first reaction, the 5′ exon–intron junction is attacked by a free hydroxyl group (provided by an internal 2′–OH position in nuclear and group II introns or by a free guanine nucleotide in group I introns). In the second reaction, the free 3′–OH at the end of the released exon in turn attacks the 3′ intron–exon junction.
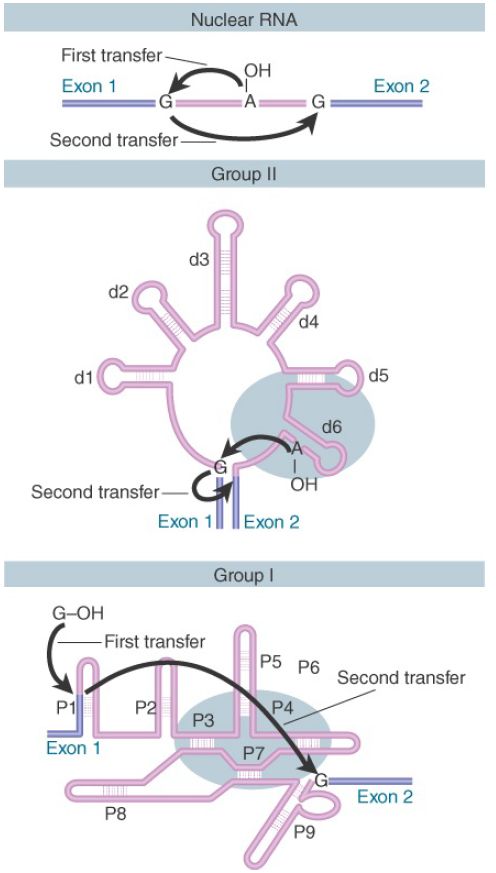
FIGURE 1 . Three classes of splicing reactions proceed by two transesterifications. First, a free –OH group attacks the exon 1–intron junction. Second, the –OH created at the end of exon 1 attacks the intron–exon 2 junction.
Parallels exist between group II introns and pre-mRNA splicing. Group II mitochondrial introns are excised by the same mechanism as nuclear pre-mRNAs via a lariat that is held together by a 2′–5′ bond. When an isolated group II RNA is incubated in vitro in the absence of additional components, it is able to perform the splicing reaction. This means that the two transesterification reactions shown in Figure 1 can be performed by the group II intron RNA sequence itself. The number of phosphodiester bonds is conserved in the reaction, and as a result an external supply of energy is not required; this could have been an important feature in the evolution of splicing.
A group II intron forms a secondary structure that contains several domains formed by base-paired stems and single-stranded loops. Domain 5 is separated by two bases from domain 6, which contains an A residue that donates the 2′–OH group for the first transesterification. This constitutes a catalytic domain in the RNA. FIGURE 2 compares this secondary structure with the structure formed by the combination of U6 with U2 and of U2 with the branch site. The similarity suggests that U6 may have a catalytic role in pre-mRNA splicing.
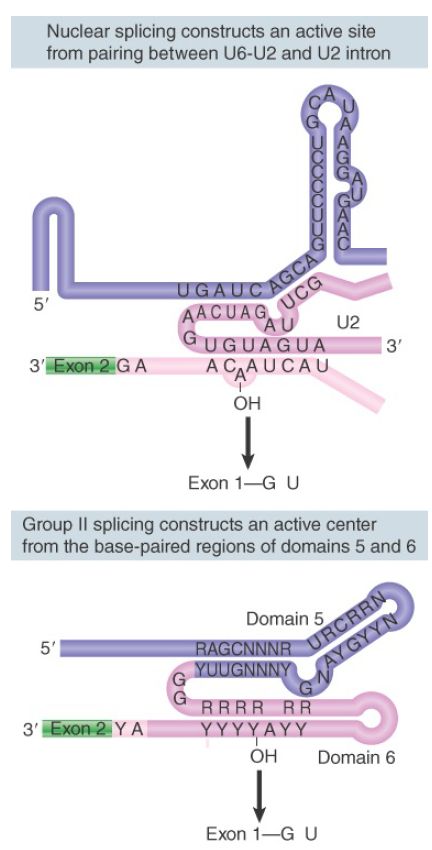
FIGURE 2. Nuclear splicing and group II splicing involve the formation of similar secondary structures. The sequences are more specific in nuclear splicing; group II splicing uses positions that may be occupied by either purine (R) or pyrimidine (Y).
The features of group II splicing suggest that splicing evolved from an autocatalytic reaction undertaken by an individual RNA molecule, in which it accomplished a controlled deletion of an internal sequence. It is likely that such a reaction would require the RNA to fold into a specific conformation, or series of conformations, and would occur exclusively in cis-conformation.
The ability of group II introns to remove themselves by an autocatalytic splicing event stands in great contrast to the requirement of nuclear introns for a complex splicing apparatus. The snRNAs of the spliceosome can be regarded as compensating for the lack of sequence information in the intron, and as providing the information required to form particular structures in RNA. The functions of the snRNAs may have evolved from the original autocatalytic system. These snRNAs act in trans upon the substrate pre-mRNA. Perhaps the ability of U1 to pair with the 5′ splice site, or of U2 to pair with the branch sequence, replaced a similar reaction that required the relevant sequence to be carried by the intron. Thus, the snRNAs may undergo reactions with the pre-mRNA substrate—and with one another—that have substituted for the series of conformational changes that occur in RNAs that splice by group II mechanisms. In effect, these changes have relieved the substrate pre-mRNA of the obligation to carry the sequences needed to sponsor the reaction. As the splicing apparatus has become more complex (and as the number of potential substrates has increased), proteins have played a more important role.

|
|
|
|
دراسة يابانية لتقليل مخاطر أمراض المواليد منخفضي الوزن
|
|
|
|
|
|
|
اكتشاف أكبر مرجان في العالم قبالة سواحل جزر سليمان
|
|
|
|
|
|
|
اتحاد كليات الطب الملكية البريطانية يشيد بالمستوى العلمي لطلبة جامعة العميد وبيئتها التعليمية
|
|
|