


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 27-4-2019
Date: 25-6-2019
Date: 3-1-2017
|
Structural formulas: Dots and dashes
For ionic compounds, the molecular formula is enough to fully identify a compound, but it’s not enough to identify covalent compounds. Look at Figure 1.1.
Both compounds have the molecular formula of C2H6O. That is, both compounds have two carbon atoms, six hydrogen atoms, and one oxygen atom. However, these are two entirely different compounds with two entirely different sets of properties. The difference is in the way the atoms are bonded, or what’s bonded to what. The compound on the left, dimethyl ether, is used in some refrigeration units and is highly flammable. The one on the right, ethyl alcohol, is the drinking variety of alcohol. Simply knowing the molecular formula isn’t enough to distinguish between the two compounds. Can you imagine going into a restaurant, ordering a shot of C2H6O, and getting dimethyl ether instead of tequila?
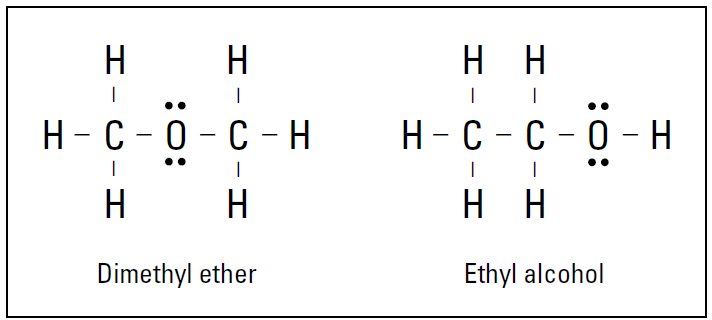
Figure 1.1: Two possible compounds of C2H6O.
Compounds that have the same molecular formula but different structures are called isomers of each other. To identify the exact covalent compound, you need its structural formula. The structural formula shows the elements in the compound, the exact number of each atom in the compound, and the bonding pattern for the compound. The electron-dot formula and Lewis formula, which I cover in this section, are common structural formulas.



|
|
|
|
4 أسباب تجعلك تضيف الزنجبيل إلى طعامك.. تعرف عليها
|
|
|
|
|
|
|
أكبر محطة للطاقة الكهرومائية في بريطانيا تستعد للانطلاق
|
|
|
|
|
|
|
العتبة العباسية المقدسة تبحث مع العتبة الحسينية المقدسة التنسيق المشترك لإقامة حفل تخرج طلبة الجامعات
|
|
|