


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
أقرأ أيضاً
التاريخ: 22-12-2016
التاريخ: 20-6-2016
التاريخ: 2023-05-29
التاريخ: 2024-05-17
|
It is a little difficult to begin at once with the present view, so we shall first see how things looked in about 1920 and then take a few things out of that picture. Before 1920, our world picture was something like this: The “stage” on which the universe goes is the three-dimensional space of geometry, as described by Euclid, and things change in a medium called time. The elements on the stage are particles, for example the atoms, which have some properties. First, the property of inertia: if a particle is moving it keeps on going in the same direction unless forces act upon it. The second element, then, is forces, which were then thought to be of two varieties: First, an enormously complicated, detailed kind of interaction force which held the various atoms in different combinations in a complicated way, which determined whether salt would dissolve faster or slower when we raise the temperature. The other force that was known was a long-range interaction—a smooth and quiet attraction—which varied inversely as the square of the distance, and was called gravitation. This law was known and was very simple. Why things remain in motion when they are moving, or why there is a law of gravitation was, of course, not known.
A description of nature is what we are concerned with here. From this point of view, then, a gas, and indeed all matter, is a myriad of moving particles. Thus, many of the things we saw while standing at the seashore can immediately be connected. First the pressure: this comes from the collisions of the atoms with the walls or whatever; the drift of the atoms, if they are all moving in one direction on the average, is wind; the random internal motions are the heat. There are waves of excess density, where too many particles have collected, and so as they rush off they push up piles of particles farther out, and so on. This wave of excess density is sound. It is a tremendous achievement to be able to understand so much. Some of these things were described in the previous chapter.
What kinds of particles are there? There were considered to be 92 at that time: 92 different kinds of atoms were ultimately discovered. They had different names associated with their chemical properties.
The next part of the problem was, what are the short-range forces? Why does carbon attract one oxygen or perhaps two oxygens, but not three oxygens? What is the machinery of interaction between atoms? Is it gravitation? The answer is no. Gravity is entirely too weak. But imagine a force analogous to gravity, varying inversely with the square of the distance, but enormously more powerful and having one difference. In gravity everything attracts everything else, but now imagine that there are two kinds of “things,” and that this new force (which is the electrical force, of course) has the property that likes repel but unlikes attract. The “thing” that carries this strong interaction is called charge.
Then what do we have? Suppose that we have two unlikes that attract each other, a plus and a minus, and that they stick very close together. Suppose we have another charge some distance away. Would it feel any attraction? It would feel practically none, because if the first two are equal in size, the attraction for the one and the repulsion for the other balance out. Therefore, there is very little force at any appreciable distance. On the other hand, if we get very close with the extra charge, attraction arises, because the repulsion of likes and attraction of unlikes will tend to bring unlikes closer together and push likes farther apart. Then the repulsion will be less than the attraction. This is the reason why the atoms, which are constituted out of plus and minus electric charges, feel very little force when they are separated by appreciable distance (aside from gravity). When they come close together, they can “see inside” each other and rearrange their charges, with the result that they have a very strong interaction. The ultimate basis of an interaction between the atoms is electrical. Since this force is so enormous, all the plusses and all minuses will normally come together in as intimate a combination as they can. All things, even ourselves, are made of fine-grained, enormously strongly interacting plus and minus parts, all neatly balanced out. Once in a while, by accident, we may rub off a few minuses or a few plusses (usually it is easier to rub off minuses), and in those circumstances we find the force of electricity unbalanced, and we can then see the effects of these electrical attractions.
To give an idea of how much stronger electricity is than gravitation, consider two grains of sand, a millimeter across, thirty meters apart. If the force between them were not balanced, if everything attracted everything else instead of likes repelling, so that there were no cancellation, how much force would there be? There would be a force of three million tons between the two! You see, there is very, very little excess or deficit of the number of negative or positive charges necessary to produce appreciable electrical effects. This is, of course, the reason why you cannot see the difference between an electrically charged or uncharged thing—so few particles are involved that they hardly make a difference in the weight or size of an object.
With this picture the atoms were easier to understand. They were thought to have a “nucleus” at the center, which is positively electrically charged and very massive, and the nucleus is surrounded by a certain number of “electrons” which are very light and negatively charged. Now we go a little ahead in our story to remark that in the nucleus itself there were found two kinds of particles, protons and neutrons, almost of the same weight and very heavy. The protons are electrically charged and the neutrons are neutral. If we have an atom with six protons inside its nucleus, and this is surrounded by six electrons (the negative particles in the ordinary world of matter are all electrons, and these are very light compared with the protons and neutrons which make nuclei), this would be atom number six in the chemical table, and it is called carbon. Atom number eight is called oxygen, etc., because the chemical properties depend upon the electrons on the outside, and in fact only upon how many electrons there are. So, the chemical properties of a substance depend only on a number, the number of electrons. (The whole list of elements of the chemists really could have been called 1, 2, 3, 4, 5, etc. Instead of saying “carbon,” we could say “element six,” meaning six electrons, but of course, when the elements were first discovered, it was not known that they could be numbered that way, and secondly, it would make everything look rather complicated. It is better to have names and symbols for these things, rather than to call everything by number.)
More was discovered about the electrical force. The natural interpretation of electrical interaction is that two objects simply attract each other: plus, against minus. However, this was discovered to be an inadequate idea to represent it. A more adequate representation of the situation is to say that the existence of the positive charge, in some sense, distorts, or creates a “condition” in space, so that when we put the negative charge in, it feels a force. This potentiality for producing a force is called an electric field. When we put an electron in an electric field, we say it is “pulled.” We then have two rules: (a) charges make a field, and (b) charges in fields have forces on them and move. The reason for this will become clear when we discuss the following phenomena: If we were to charge a body, say a comb, electrically, and then place a charged piece of paper at a distance and move the comb back and forth, the paper will respond by always pointing to the comb. If we shake it faster, it will be discovered that the paper is a little behind, there is a delay in the action. (At the first stage, when we move the comb rather slowly, we find a complication which is magnetism. Magnetic influences have to do with charges in relative motion, so magnetic forces and electric forces can really be attributed to one field, as two different aspects of exactly the same thing. A changing electric field cannot exist without magnetism.) If we move the charged paper farther out, the delay is greater. Then an interesting thing is observed. Although the forces between two charged objects should go inversely as the square of the distance, it is found, when we shake a charge, that the influence extends very much farther out than we would guess at first sight. That is, the effect falls off more slowly than the inverse square.
Here is an analogy: If we are in a pool of water and there is a floating cork very close by, we can move it “directly” by pushing the water with another cork. If you looked only at the two corks, all you would see would be that one moved immediately in response to the motion of the other—there is some kind of “interaction” between them. Of course, what we really do is to disturb the water; the water then disturbs the other cork. We could make up a “law” that if you pushed the water a little bit, an object close by in the water would move. If it were farther away, of course, the second cork would scarcely move, for we move the water locally. On the other hand, if we jiggle the cork a new phenomenon is involved, in which the motion of the water moves the water there, etc., and waves travel away, so that by jiggling, there is an influence very much farther out, an oscillatory influence, that cannot be understood from the direct interaction. Therefore, the idea of direct interaction must be replaced with the existence of the water, or in the electrical case, with what we call the electromagnetic field.
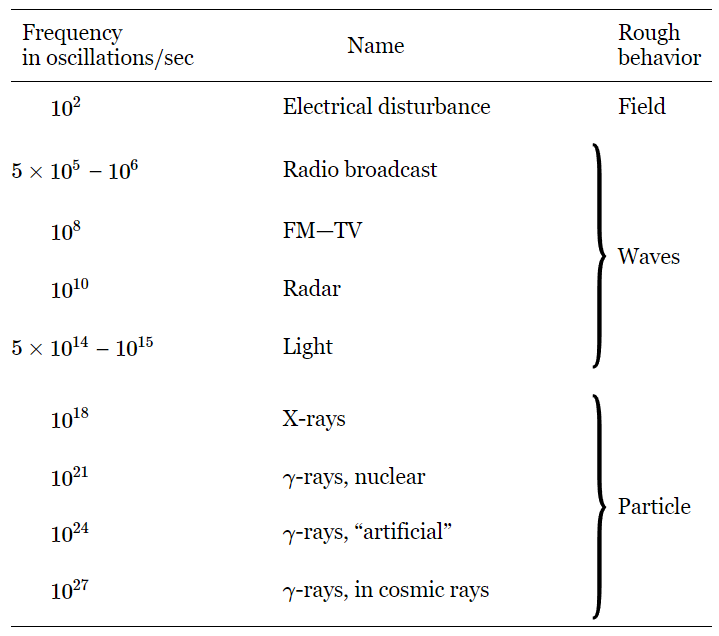
The electromagnetic field can carry waves; some of these waves are light, others are used in radio broadcasts, but the general name is electromagnetic waves. These oscillatory waves can have various frequencies. The only thing that is really different from one wave to another is the frequency of oscillation. If we shake a charge back and forth more and more rapidly, and look at the effects, we get a whole series of different kinds of effects, which are all unified by specifying but one number, the number of oscillations per second. The usual “pickup” that we get from electric currents in the circuits in the walls of a building have a frequency of about one hundred cycles per second. If we increase the frequency to 500 or 1000 kilocycles (1 kilocycle=1000 cycles) per second, we are “on the air,” for this is the frequency range which is used for radio broadcasts. (Of course, it has nothing to do with the air! We can have radio broadcasts without any air.) If we again increase the frequency, we come into the range that is used for FM and TV. Going still further, we use certain short waves, for example for radar. Still higher, and we do not need an instrument to “see” the stuff, we can see it with the human eye. In the range of frequency from 5×1014 to 1015 cycles per second our eyes would see the oscillation of the charged comb, if we could shake it that fast, as red, blue, or violet light, depending on the frequency. Frequencies below this range are called infrared, and above it, ultraviolet. The fact that we can see in a particular frequency range makes that part of the electromagnetic spectrum no more impressive than the other parts from a physicist’s standpoint, but from a human standpoint, of course, it is more interesting. If we go up even higher in frequency, we get x-rays. X-rays are nothing but very high-frequency light. If we go still higher, we get gamma rays. These two terms, x-rays and gamma rays, are used almost synonymously. Usually, electromagnetic rays coming from nuclei are called gamma rays, while those of high energy from atoms are called x-rays, but at the same frequency they are indistinguishable physically, no matter what their source. If we go to still higher frequencies, say to 1024 cycles per second, we find that we can make those waves artificially, for example with the synchrotron here at Caltech. We can find electromagnetic waves with stupendously high frequencies—with even a thousand times more rapid oscillation—in the waves found in cosmic rays. These waves cannot be controlled by us.
Table 2–1
The Electromagnetic Spectrum



|
|
|
|
للتخلص من الإمساك.. فاكهة واحدة لها مفعول سحري
|
|
|
|
|
|
|
العلماء ينجحون لأول مرة في إنشاء حبل شوكي بشري وظيفي في المختبر
|
|
|
|
|
|
|
قسم العلاقات العامّة ينظّم برنامجاً ثقافياً لوفد من أكاديمية العميد لرعاية المواهب
|
|
|