


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 23-10-2019
Date: 12-12-2020
Date: 20-8-2019
|
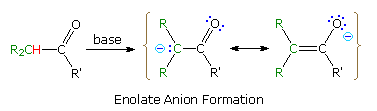
Many carbon acids have enhanced acidity because of a neighboring functional group. The acidity of alpha hydrogens in aldehydes, ketones and esters is well documented, and is the source of many important synthetic procedures. The following equation illustrates the general enolate anion transformation, with the acidic alpha-hydrogen colored red. The resulting ambident anion is stabilized by charge delocalization, and may react with electrophiles at both carbon and oxygen.
Stereoelectronic factors govern the enolization reaction, as illustrated by clicking on the diagram below. The bond from the alpha carbon to the acidic alpha-hydrogen must be oriented 90º to the plane of the carbonyl group, or parallel to the pi-electron system (colored magenta here). The ideal overlap occurs with a 0º dihedral angle between this bond and the pi-orbital, as shown.
By clicking on the diagram a second time, the importance of this stereoelectronic requirement will be demonstrated. An increase in the acidity of carbon acids activated by two carbonyl groups is well known, and is illustrated by the two beta-dicarbonyl compounds on the left side of the diagram. In such cases the acidic C-H unit may be oriented perpendicular to both carbonyl groups, and the resulting planar anion is stabilized by additional charge delocalization (over both oxygens and the central carbon). In the case of the bicyclic diketone on the right, the C-H bond nearly eclipses the two carbonyl C-O bonds, resulting in a dihedral angle with the pi-electron systems of roughly 90º. Consequently, the acidity of this hydrogen is similar to that of the hydrogens of an alkane or cycloalkane. It should also be apparent that if an enolate anion were to be formed to the bridgehead carbon, the double bond would be prohibited by Bredt's rule.



|
|
|
|
دراسة يابانية لتقليل مخاطر أمراض المواليد منخفضي الوزن
|
|
|
|
|
|
|
اكتشاف أكبر مرجان في العالم قبالة سواحل جزر سليمان
|
|
|
|
|
|
|
اتحاد كليات الطب الملكية البريطانية يشيد بالمستوى العلمي لطلبة جامعة العميد وبيئتها التعليمية
|
|
|