


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 5-4-2016
Date: 24-6-2017
Date: 24-4-2017
|
The Mole
The mole (abbreviated mol) is the SI unit for the amount of a chemical substance. It is always associated with specific microscopic entities such as atoms, molecules, ions, electrons, other particles, or specified groups of such particles as represented by a chemical formula. It is the amount of the specified substance that contains the same number of particles as the number of carbon atoms in exactly 12 grams of 12C. This important number is Avogadro’s number NA=6.022 x 1023. The molar mass M of a substance is the mass in grams of 1 mole of that substance. We calculate molar masses by summing the atomic masses of all the atoms appearing in a chemical formula. For example, the molar mass of formaldehyde CH2O is Thus, 1 mole of formaldehyde has a mass of 30.0 g, and 1 mole of glucose has a mass of 180.0 g.
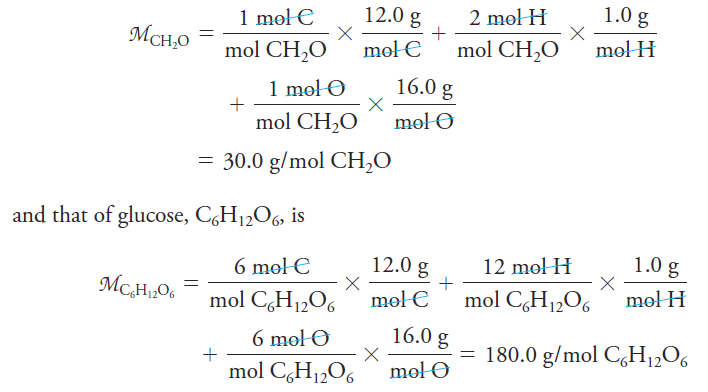



|
|
|
|
التوتر والسرطان.. علماء يحذرون من "صلة خطيرة"
|
|
|
|
|
|
|
مرآة السيارة: مدى دقة عكسها للصورة الصحيحة
|
|
|
|
|
|
|
العتبة العباسية المقدسة تُطلق فعّاليات مؤتمر ذاكرة الألم في العراق
|
|
|