


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 10-9-2017
Date: 6-4-2016
Date: 11-9-2017
|
BUTADIENE (CH2=CH-CH=CH2)
Butadiene is by far the most important monomer for synthetic rubber production. It can be polymerized to polybutadiene or copolymerized with styrene to styrene-butadiene rubber (SBR). Butadiene is an important intermediate for the synthesis of many chemicals such as hexamethylenediamine and adipic acid. Both are monomers for producing nylon. Chloroprene is another butadiene derivative for the synthesis of neoprene rubber.
The unique role of butadiene among other conjugated diolefins lies in its high reactivity as well as its low cost. Butadiene is obtained mainly as a coproduct with other light olefins from steam cracking units for ethylene production. Other sources of butadiene are the catalytic dehydrogenation of butanes and butenes, and dehydration of 1,4-butanediol. Butadiene is a colorless gas with a mild aromatic odor. Its specific gravity is 0.6211 at 20°C and its boiling temperature is –4.4°C. The U.S. production of butadiene reached 4.1 billion pounds in 1997 and it was the 36th highest-volume chemical.
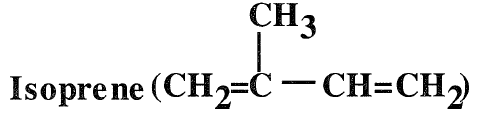
Isoprene (2-methyl-1,3-butadiene) is a colorless liquid, soluble in alcohol but not in water. Its boiling temperature is 34.1°C. Isoprene is the second important conjugated diene for synthetic rubber production. The main source for isoprene is the dehydrogenation of C5 olefins (tertiary amylenes) obtained by the extraction of a C5 fraction from catalytic cracking units. It can also be produced through several synthetic routes using reactive chemicals such as isobutene, formaldehyde, and propene . The main use of isoprene is the production of polyisoprene. It is also a comonomer with isobutene for butyl rubber production.



|
|
|
|
للعاملين في الليل.. حيلة صحية تجنبكم خطر هذا النوع من العمل
|
|
|
|
|
|
|
"ناسا" تحتفي برائد الفضاء السوفياتي يوري غاغارين
|
|
|
|
|
|
|
نحو شراكة وطنية متكاملة.. الأمين العام للعتبة الحسينية يبحث مع وكيل وزارة الخارجية آفاق التعاون المؤسسي
|
|
|