


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 21-8-2016
Date: 18-1-2020
Date: 23-5-2017
|
Naming Alkanes
In earlier times, when relatively few pure organic chemicals were known, new compounds were named at the whim of their discoverer. Thus, urea (CH4N2O) is a crystalline substance isolated from urine; morphine (C17H19NO3) is an analgesic (painkiller) named after Morpheus, the Greek god of dreams; and acetic acid, the primary organic constituent of vinegar, is named from the Latin word for vinegar, acetum. As the science of organic chemistry slowly grew in the 19th century, so too did the number of known compounds and the need for a systematic method of naming them. The system of nomenclature we’ll use in this book is that devised by the International Union of Pure and Applied Chemistry (IUPAC, usually pronounced as eye-you-pac). A chemical name typically has four parts in the IUPAC system of nomenclature: prefix, parent, locant, and suffix. The prefix identifies the various substituent groups in the molecule, the parent selects a main part of the molecule and tells how many carbon atoms are in that part, the locants give the positions of the functional groups and substituents, and the suffix identifies the primary functional group.

As we cover new functional groups in later chapters, the applicable IUPAC rules of nomenclature will be given. In addition, Appendix A at the back of this book gives an overall view of organic nomenclature and shows how compounds that contain more than one functional group are named. (If preferred, you can study that appendix now.) For the present, let’s see how to name branched-chain alkanes and learn some general rules that are applicable to all compounds.
All but the most complex branched-chain alkanes can be named by following four steps. For a very few compounds, a fifth step is needed.
Step 1
Find the parent hydrocarbon.
(a) Find the longest continuous chain of carbon atoms in the molecule, and use the name of that chain as the parent name. The longest chain may not always be apparent from the manner of writing; you may have to “turn corners.”
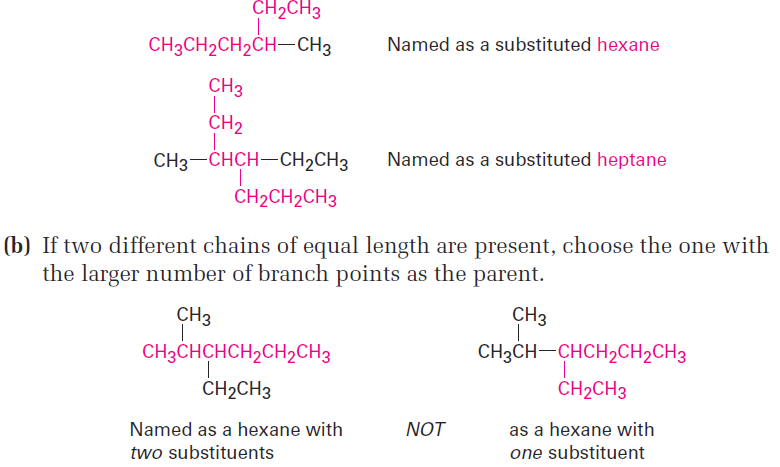
Step 2
Number the atoms in the longest chain.
(a) Beginning at the end nearer the first branch point, number each carbon atom in the parent chain.

The first branch occurs at C3 in the proper system of numbering, not at C4.
(b) If there is branching an equal distance away from both ends of the parent chain, begin numbering at the end nearer the second branch point.

Step 3
Identify and number the substituents.
(a) Assign a number, or locant, to each substituent to locate its point of attachment to the parent chain.
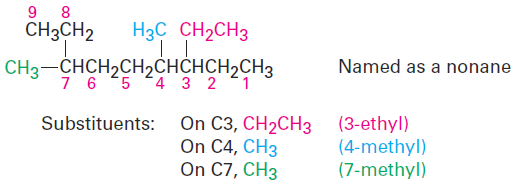
(b) If there are two substituents on the same carbon, give both the same number. There must be as many numbers in the name as there are substituents.
Step 4
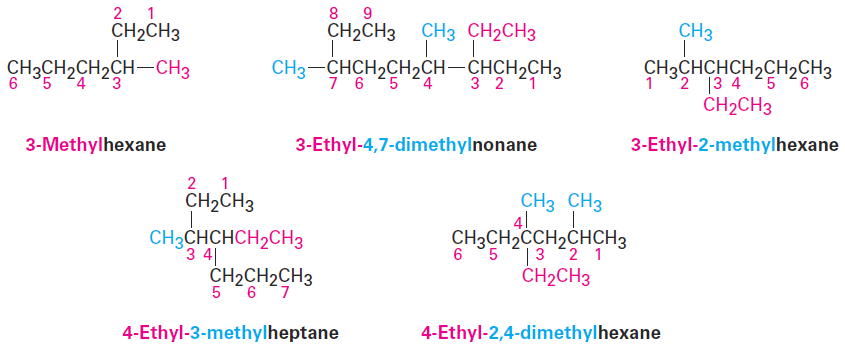
Write the name as a single word.
Use hyphens to separate the different prefixes, and use commas to separate numbers. If two or more different substituents are present, cite them in alphabetical order. If two or more identical substituents are present on the parent chain, use one of the multiplier prefixes di-, tri-, tetra-, and so forth, but don’t use these prefixes for alphabetizing. Full names for some of the examples we have been using are as follows:
Step 5
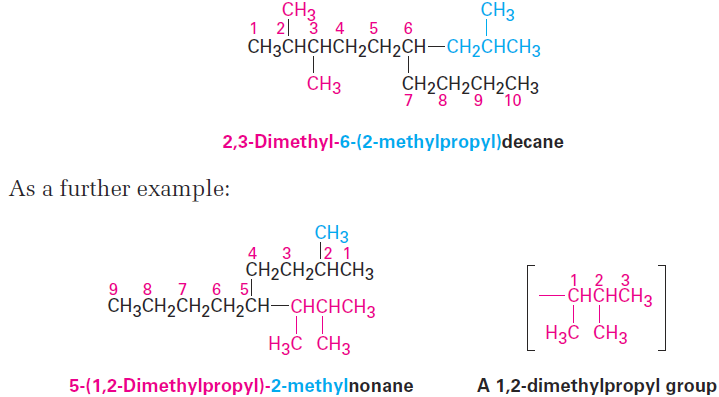
Name a branched substituent as though it were itself a compound. In some particularly complex cases, a fifth step is necessary. It occasionally happens that a substituent on the main chain is itself branched. In the following case, for instance, the substituent at C6 is a three-carbon chain with a methyl group. To name the compound fully, the branched substituent must first be named.
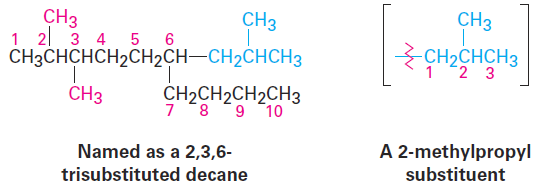
Number the branched substituent beginning at the point of its attachment to the main chain, and identify it—in this case, a 2-methylpropyl group. The substituent is treated as a whole and is alphabetized according to the first letter of its complete name, including any numerical prefix. It is set off in parentheses when naming the entire molecule.
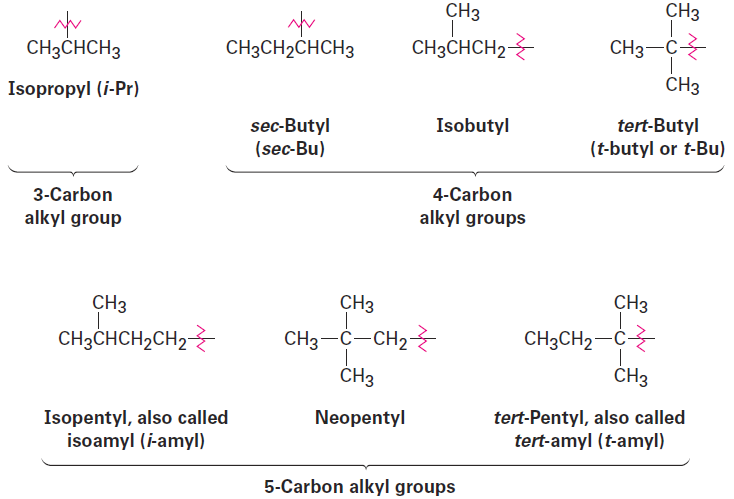
For historical reasons, some of the simpler branched-chain alkyl groups also have nonsystematic, common names, as noted earlier.
The common names of these simple alkyl groups are so well entrenched in the chemical literature that IUPAC rules make allowance for them. Thus, the following compound is properly named either 4-(1-methylethyl)heptane or 4-isopropylheptane. There’s no choice but to memorize these common names; fortunately, there are only a few of them.
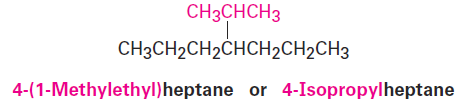
When writing an alkane name, the nonhyphenated prefix iso- is considered part of the alkyl-group name for alphabetizing purposes, but the hyphenated and italicized prefixes sec- and tert- are not. Thus, isopropyl and isobutyl are listed alphabetically under i, but sec-butyl and tert-butyl are listed under b.



|
|
|
|
التوتر والسرطان.. علماء يحذرون من "صلة خطيرة"
|
|
|
|
|
|
|
مرآة السيارة: مدى دقة عكسها للصورة الصحيحة
|
|
|
|
|
|
|
نحو شراكة وطنية متكاملة.. الأمين العام للعتبة الحسينية يبحث مع وكيل وزارة الخارجية آفاق التعاون المؤسسي
|
|
|