
تاريخ الفيزياء

علماء الفيزياء


الفيزياء الكلاسيكية

الميكانيك

الديناميكا الحرارية


الكهربائية والمغناطيسية

الكهربائية

المغناطيسية

الكهرومغناطيسية


علم البصريات

تاريخ علم البصريات

الضوء

مواضيع عامة في علم البصريات

الصوت


الفيزياء الحديثة


النظرية النسبية

النظرية النسبية الخاصة

النظرية النسبية العامة

مواضيع عامة في النظرية النسبية

ميكانيكا الكم

الفيزياء الذرية

الفيزياء الجزيئية


الفيزياء النووية

مواضيع عامة في الفيزياء النووية

النشاط الاشعاعي


فيزياء الحالة الصلبة

الموصلات

أشباه الموصلات

العوازل

مواضيع عامة في الفيزياء الصلبة

فيزياء الجوامد


الليزر

أنواع الليزر

بعض تطبيقات الليزر

مواضيع عامة في الليزر


علم الفلك

تاريخ وعلماء علم الفلك

الثقوب السوداء


المجموعة الشمسية

الشمس

كوكب عطارد

كوكب الزهرة

كوكب الأرض

كوكب المريخ

كوكب المشتري

كوكب زحل

كوكب أورانوس

كوكب نبتون

كوكب بلوتو

القمر

كواكب ومواضيع اخرى

مواضيع عامة في علم الفلك

النجوم

البلازما

الألكترونيات

خواص المادة


الطاقة البديلة

الطاقة الشمسية

مواضيع عامة في الطاقة البديلة

المد والجزر

فيزياء الجسيمات


الفيزياء والعلوم الأخرى

الفيزياء الكيميائية

الفيزياء الرياضية

الفيزياء الحيوية

الفيزياء والفلسفة

الفيزياء العامة


مواضيع عامة في الفيزياء

تجارب فيزيائية

مصطلحات وتعاريف فيزيائية

وحدات القياس الفيزيائية

طرائف الفيزياء

مواضيع اخرى
THAWING AND FREEZING
المؤلف:
S. Gibilisco
المصدر:
Physics Demystified
الجزء والصفحة:
p278
28-9-2020
1939
THAWING AND FREEZING
Consider our old friend, water. Imagine that it is late winter in a place such as northern Wisconsin and that the temperature of the water ice on the lake is exactly 0°C. The ice is not safe to skate on, as it was in the middle of the winter, because the ice has become “soft.” It is more like slush than ice. It is partly solid and partly liquid. Nevertheless, the temperature of this soft ice is 0°C.
As the temperature continues to rise, the slush gets softer. It becomes proportionately more liquid water and less solid ice. However, its temperature remains at 0°C. Eventually, all the ice melts into liquid. This can take place with astonishing rapidity. You might leave for school one morning and see the lake nearly “socked in” with slush and return in the evening to find it almost entirely thawed. Now you can get the canoe out! But you won’t want to go swimming. The liquid water will stay at 0°C until all the ice is gone. Only then will the temperature begin to rise slowly. Consider now what happens in the late autumn. The weather, and the water, is growing colder. The water finally drops to 0°C. The surface begins to freeze. The temperature of this new ice is 0°C. Freezing takes place until the whole lake surface is solid ice. The weather keeps growing colder (a lot colder if you live in northern Wisconsin). Once the surface is entirely solid ice, the temperature of the ice begins to fall below 0°C, although it remains at 0°C at the boundary just beneath the surface where solid ice meets liquid water. The layer of ice gets thicker. The ice near the surface can get much colder than 0°C. How much colder depends on various factors, such as the severity of the winter and the amount of snow that happens to fall on top of the ice and insulate it against the bitter chill of the air.
The temperature of water does not follow exactly along with the air temperature when heating or cooling takes place in the vicinity of 0°C. Instead, the water temperature follows a curve something like that shown in Fig. 1. In part a, the air temperature is getting warmer; in part b, it is getting colder. The water “stalls” as it thaws or freezes. Other substances exhibit this same property when they thaw or freeze.
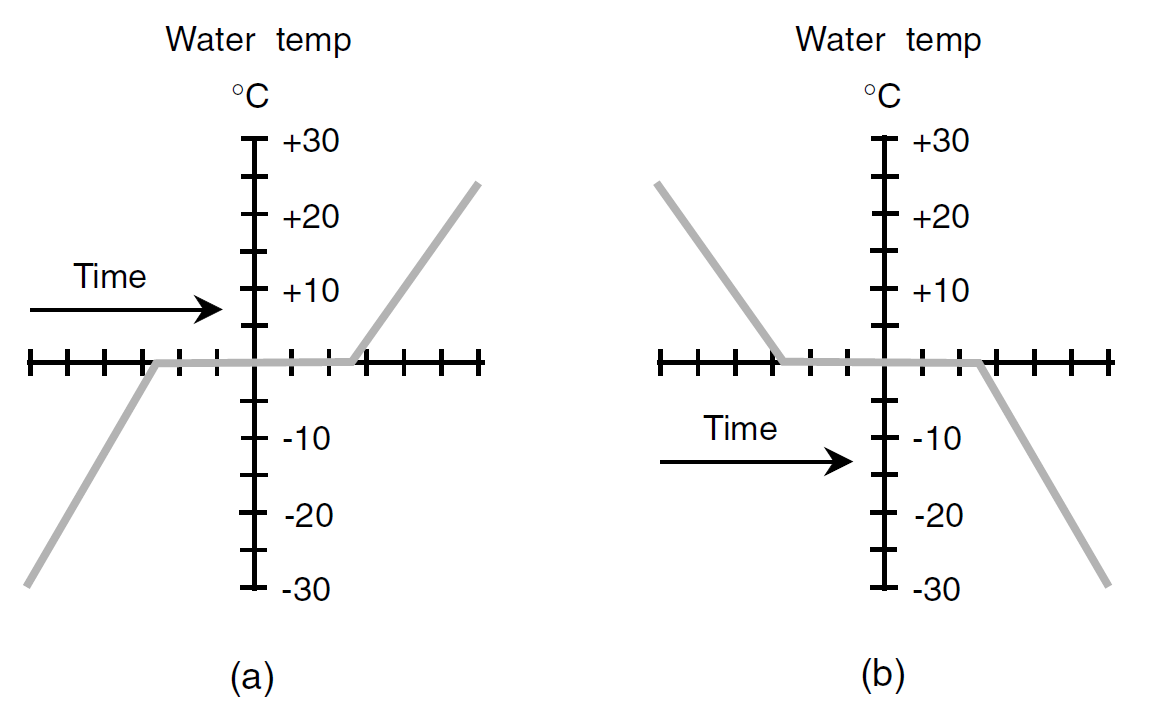
Fig. 1. Water as it thaws and freezes. (a) The environmental temperature is getting warmer, and the ice is thawing. (b) The environmental temperature is getting colder, and the liquid water is freezing.
 الاكثر قراءة في الفيزياء العامة
الاكثر قراءة في الفيزياء العامة
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















