


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 24-10-2019
Date: 3-12-2015
Date: 7-10-2020
|
It has been noted that dipolar phosphorus compounds are stabilized by p-d bonding. This bonding stabilization extends to carbanions adjacent to phosphonium centers, and the zwitterionic conjugate bases derived from such cations are known as ylides. An ylide is defined as a compound with opposite charges on adjacent atoms both of which have complete octets. For the Wittig reaction discussed below an organophosphorus ylide, also called Wittig reagents, will be used. The ability of phosphorus to hold more than eight valence electrons allows for a resonance structure to be drawn forming a double bonded structure.
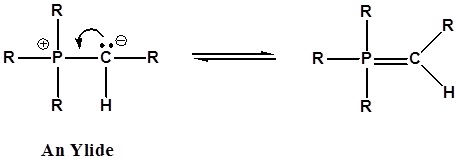
The stabilization of the carbanion provided by the phosphorus causes an increase in acidity (pKa ~35). Very strong bases, such as butyl lithium, are required for complete formation of ylides.
The ylides shown here are all strong bases. Like other strongly basic organic reagents, they are protonated by water and alcohols, and are sensitive to oxygen. Water decomposes phosphorous ylides to hydrocarbons and phosphine oxides, as shown.
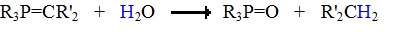
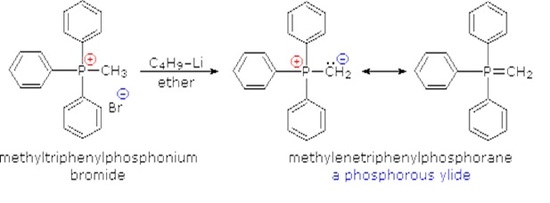
Although many ylides are commercially available it is often necessary to create them synthetically. Ylides can be synthesized from an alkyl halide and a trialkyl phosphine. Typically triphenyl phosphine is used to synthesize ylides. Because a SN2 reaction is used in the ylide synthesis methyl and primary halides perform the best. Secondary halides can also be used but the yields are generally lower. This should be considered when planning out a synthesis which involves a synthesized Wittig reagent.




|
|
|
|
"إنقاص الوزن".. مشروب تقليدي قد يتفوق على حقن "أوزيمبيك"
|
|
|
|
|
|
|
الصين تحقق اختراقا بطائرة مسيرة مزودة بالذكاء الاصطناعي
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|