The Joule experiment
When there are no interactions between the molecules, the internal energy is independent of their separation and hence independent of the volume of the sample (see Molecular interpretation 2.2). Therefore, for a perfect gas we can write πT = 0. The statement πT = 0 (that is, the internal energy is independent of the volume occupied by the sample) can be taken to be the definition of a perfect gas, for later we shall see that it implies the equation of state pV = nRT. If the internal energy increases (dU > 0) as the volume of the sample expands isothermally (dV > 0), which is the case when there are attractive forces between the particles, then a plot of internal energy against volume slopes upwards and πT > 0 (Fig. 2.25). James Joule thought that he could measure πT by observing the change in temperature of a gas when it is allowed to expand into a vacuum. He used two metal vessels immersed in a water bath (Fig. 2.26). One was filled with air at about 22 atm and the other was evacuated. He then tried to measure the change in temperature of the water of the bath when a stopcock was opened and the air expanded into a vacuum. He observed no change in temperature.
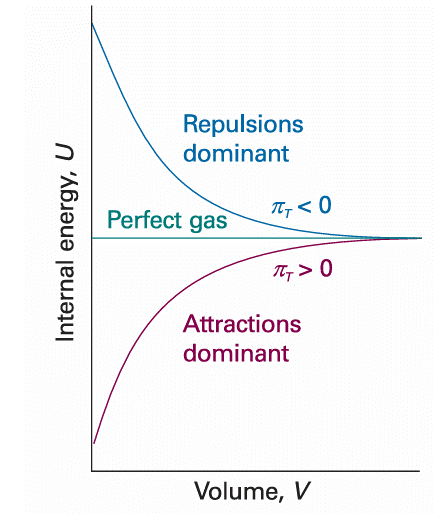
Fig. 2.25 For a perfect gas, the internal energy is independent of the volume (at constant temperature). If attractions are dominant in a real gas, the internal energy increases with volume because the molecules become farther apart on average. If repulsions are dominant, the internal energy decreases as the gas expands.
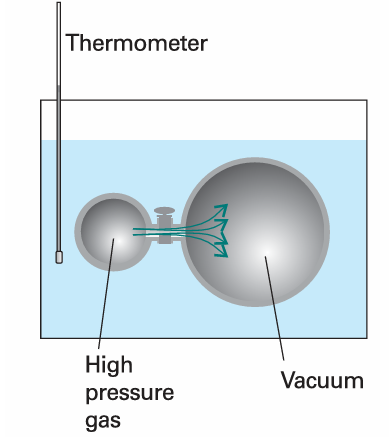
Fig. 2.26 A schematic diagram of the apparatus used by Joule in an attempt to measure the change in internal energy when a gas expands isothermally. The heat absorbed by the gas is proportional to the change in temperature of the bath.
The thermodynamic implications of the experiment are as follows. No work was done in the expansion into a vacuum, so w = 0. No energy entered or left the system (the gas) as heat because the temperature of the bath did not change, so q = 0. Consequently, within the accuracy of the experiment, ∆U = 0. It follows that U does not change much when a gas expands isothermally and therefore that πT = 0. Joule’s experiment was crude. In particular, the heat capacity of the apparatus was so large that the temperature change that gases do in fact cause was too small to measure. From his experiment Joule extracted an essential limiting property of a gas, a property of a perfect gas, without detecting the small deviations characteristic of real gases.
