

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء الاشعاعية والنووية
Directions for Filtering and Igniting Precipitates
المؤلف:
D. A. Skoog, F. J.Holler, D M. West, and S. R. Crouch
المصدر:
Fundamentals of Analytical Chemistry
الجزء والصفحة:
9th p32
27-3-2016
6957
Directions for Filtering and Igniting Precipitates
Preparation of a Filter Paper
Figure 1 shows the sequence for folding and seating a filter paper in a 60-deg funnel. The paper is folded exactly in half (a), firmly creased, and folded again (b). A triangular piece from one of the corners is torn off parallel to the second fold (c). The paper is then opened so that the untorn quarter forms a cone (d).

Figure 1: Folding and seating a filter paper. (a) Fold the paper exactly in half and crease it firmly. (b) Fold the paper a second time. (c) Tear off one of the corners on a line parallel to the second fold. (d) Open the untorn half of the folded paper to form a cone. (e) Seat the cone firmly into the funnel. (f) Moisten the paper slightly and gently pat the paper into place.
The cone is fitted into the funnel, and the second fold is creased (e). Seating is completed by dampening the cone with water from a wash bottle and gently patting it with a finger (f ). There will be no leakage of air between the funnel and a properly seated cone. In addition, the stem of the funnel will be filled with an unbroken column of liquid.
Transferring Paper and Precipitate to a Crucible
After filtration and washing have been completed, the filter and its contents must be transferred from the funnel to a crucible that has been brought to constant mass. Ashless paper has very low wet strength and must be handled with care during the transfer. The danger of tearing is lessened considerably if the paper is allowed to dry somewhat before it is removed from the funnel.

Figure 2 Transferring a filter paper and precipitate from a funnel to a crucible. (a) Pull the triple-thick portion of the cone to the opposite side of the funnel. (b) Remove the filter cone from the funnel, and flatten the cone along its upper edge. (c) Fold the corners inward. (d) Fold the top edge of the cone toward the tip to enclose the precipitate in the paper. (e) Gently ease the folded paper and its contents into the crucible.
Figure 2 illustrates the transfer process. The triple-thick portion of the filter paper is drawn across the funnel (a) to flatten the cone along its upper edge (b); the corners are next folded inward (c); and the top edge is then folded over (d). Finally, the paper and its contents are eased into the crucible (e) so that the bulk of the precipitate is near the bottom.
Ashing Filter Papers
If a heat lamp is used, the crucible is placed on a clean, nonreactive surface, such as a wire screen covered with aluminum foil. The lamp is then positioned about 1 cm above the rim of the crucible and turned on. Charring takes place without further attention. The process is considerably accelerated if the paper is moistened with no more than one drop of concentrated ammonium nitrate solution. The residual carbon is eliminated with a burner, as described in the next paragraph. Considerably more attention must be paid if a burner is used to ash a filter paper because the burner produces much higher temperatures than a heat lamp. Thus, mechanical loss of precipitate may occur if moisture is expelled too rapidly in the initial stages of heating or if the paper bursts into flame. Also, partial reduction of some precipitates can occur through reaction with the hot carbon of the charring paper. This reduction is a serious problem if reoxidation following ashing is inconvenient. These difficulties can be minimized by positioning the crucible as illustrated in Figure 3.
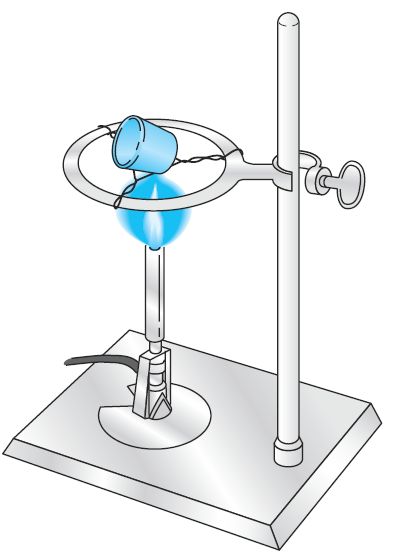
Figure 3 Ignition of a precipitate. Proper crucible position for preliminary charring is shown.
The tilted position allows for the easy access of air. A clean crucible cover should be kept handy to extinguish any flame. Heating should begin with a small flame. The temperature is gradually increased as moisture is evolved and the paper begins to char. The amount of smoke given off indicates the intensity of heating that can be tolerated. Thin wisps are normal. A significant increase in smoke indicates that the paper is about to flash and that heating should be temporarily discontinued. Any flame should be immediately extinguished with a crucible cover. (The cover may become discolored from the condensation of carbonaceous products. These products must ultimately be removed from the cover by ignition to confirm the absence of entrained particles of precipitate.) When no further smoking can be detected, heating is increased to eliminate the residual carbon.
Strong heating, as necessary, can then be undertaken. This sequence usually precedes the final ignition of a precipitate in a muffle furnace, where a reducing atmosphere is equally undesirable.
Using Filtering Crucibles
A vacuum filtration train (Figure 4) is used when a filtering crucible can be used instead of paper. The trap isolates the filter flask from the source of vacuum.
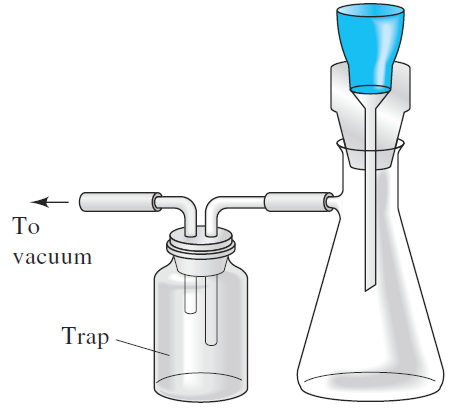
Figure 4 Train for vacuum filtration. The trap isolates the filter flask from the source of vacuum.
 الاكثر قراءة في طرق الفصل والتنقية
الاكثر قراءة في طرق الفصل والتنقية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















