Calorimetry
Calorimetry is the study of heat transfer during physical and chemical processes. A calorimeter is a device for measuring energy transferred as heat. The most common device for measuring ∆U is an adiabatic bomb calorimeter (Fig. 2.9). The process we wish to study—which may be a chemical reaction—is initiated inside a constant volume container, the ‘bomb’. The bomb is immersed in a stirred water bath, and the whole device is the calorimeter. The calorimeter is also immersed in an outer water bath. The water in the calorimeter and of the outer bath are both monitored and adjusted to the same temperature. This arrangement ensures that there is no net loss of heat from the calorimeter to the surroundings (the bath) and hence that the calorimeter is adiabatic. The change in temperature, ∆T, of the calorimeter is proportional to the heat that the reaction releases or absorbs. Therefore, by measuring ∆T we can determine qV and hence find ∆U. The conversion of ∆T to qV is best achieved by calibrating the calorimeter using a process of known energy output and determining the calorimeter constant, the constant C in the relation , q =C∆T , The calorimeter constant may be measured electrically by passing a constant current, I, from a source of known potential difference, V, through a heater for a known period of time, t, for then , q =IVt , Alternatively, C may be determined by burning a known mass of substance (benzoic acid is often used) that has a known heat output. With C known, it is simple to interpret an observed temperature rise as a release of heat.

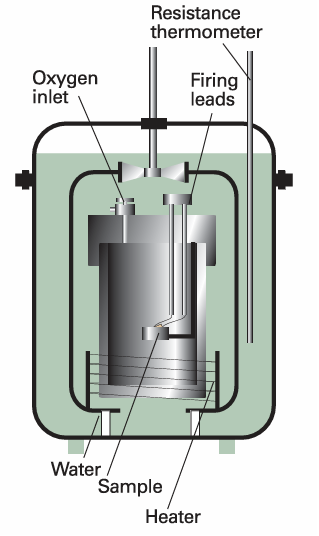
Fig. 2.9 A constant-volume bomb calorimeter. The ‘bomb’ is the central vessel, which is strong enough to withstand high pressures. The calorimeter (for which the heat capacity must be known) is the entire assembly shown here. To ensure adiabaticity, the calorimeter is immersed in a water bath with a temperature continuously readjusted to that of the calorimeter at each stage of the combustion.
