


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 21-4-2016
Date: 2-11-2020
Date: 6-12-2015
|
Cell Adhesion Molecules
Cell adhesion molecules play central roles in embryonic development, tissue organization, and overall maintenance of the structure and form of multicellular organisms (1, 2) They provide the physical links between cells and other cells, and between cells and the extracellular matrix. Adhesion molecules are also involved in dynamic cell interactions, such as during tissue morphogenesis, cell migration, immunological and inflaemmatory responses, and possibly memory formation (1, 3-7). Adhesive proteins are also intimately involved in intracellular signaling processes that regulate cell growth, differentiation, gene regulation, and prograemmed cell death (8-13).
Cell adhesion molecules mediate adhesive interactions by forming specific protein-protein interactions or between proteins and complex carbohydrates. In addition, cell adhesion molecules frequently link directly to multimolecular protein complexes on the cytoplasmic face of the plasma membrane, which, in turn, mediate interactions with the cytoskeleton and signal transduction pathways (3, 9-13). Consequently, these cell adhesion and signaling complexes not only link cells with other cells and the extracellular matrix but also help to integrate extracellular physical information with the major signal transduction pathways within cells. For example, they appear to transduce information from inputs as diverse as transient contacts with other cells, binding of different connective tissue molecules, and tension or torsion at the cell surface. Each of these inputs connects to intracellular signaling pathways, such as phosphorylation cascades (9-12).
1. Types of Adhesion
The physical linkages involved in cell adhesion are provided by adhesion molecules that function as parts of general adhesion systems or in specialized adhesive structures. For example, epithelial cells can adhere to other cells along broad expanses of plasma membrane using general-purpose adhesive molecules such as cadherins (14-16). They can also adhere to each other by specialized adhesive structures, such as adherens junctions, desmosomes, and tight junctions (1). Each type of specialized
junctional complex requires specific adhesion molecule components, which can include specialized cadherins and other proteins. Fibroblasts can also form complexes with other cells, but they most characteristically adhere to extracellular matrix. Cell-to-matrix adhesions can be broad and flat, as when epithelial cells adhere via integrins to basement membranes, or they can involve specialized structures that include hemidesmosomes or the focal contacts of fibroblastic and endothelial cells. The protein complexes involved in cell-to-matrix adhesion contain different complements of adhesive, cytoskeletal, and signal transduction molecules than those involved in cell-to-cell adhesion (1, 13, 15). This entry will first provide a general overview of specific types of cell adhesion proteins, then briefly discuss how they are integrated into cytoskeletal and signaling networks.
2. General Features of Cell Adhesion Proteins
Cell adhesiveness is generally based on the specific binding of a protein to another molecule at the cell surface. When adhesion results from binding of an adhesion molecule to the same type of protein on a neighboring cell, the interaction is termed ‘homophilic.’ The cadherin family is a major mediator of such homophilic interactions (14-16) (see Cadherins). Cadherins form complexes in which both intercellular and lateral binding interactions cooperate to create tightly packed adhesion complexes that mediate adhesion with high avidity.
In many systems, however, adhesiveness involves binding of a receptor to a specific ligand. In cell-to-cell adhesion, the target protein of an adhesion receptor can be a “counter-receptor” or a complex carbohydrate on a protein anchor in the plasma membrane. In cell-to-matrix interactions, a plasma membrane adhesion protein such as an integrin can bind to an extracellular matrix protein that is itself considered to be an adhesive protein. For example, the protein bound could be a fibronectin or a laminin, which are complex, multifunctional proteins involved in both cell adhesion and anchorage to structural components of the extracellular matrix (2, 15, 17).
Consequently, there are two broad classes of adhesion molecules or receptors (Fig. 1). One class is bound to the plasma membrane, often as a transmembrane protein (Fig. 1). This type of molecule is generally a receptor, a homophilic adhesion molecule, or a counterreceptor. It often consists of an extracellular domain containing one or more cell-interaction domains or sites, as well as a stalk region, a hydrophobic transmembrane domain, and (usually) a cytoplasmic domain or tail. This type of molecule is often also involved in transmembrane transmission of signals.
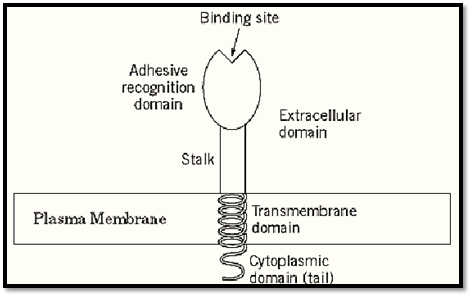
Figure 1. Schematic diagram of a generalized transmembrane adhesion molecule consisting of an extracellular domain, transmembrane segment, and cytoplasmic domain.
The second broad class of adhesion molecules consists of proteins that are often classified as cell surface or extracellular matrix proteins, but that contain domains or sites involved in cellular adhesion (Fig. 2). Molecules in this class include fibronectins, laminins, vitronectin, tenascins, thrombospondins, and the collagens. All contain one or more cell-binding domains, which consist of a primary recognition motif consisting of a short peptide sequence (eg, Arg—Gly—Asp), and sometimes a synergy site that provides a substantial increase in receptor-binding specificity and affinity. As discussed below, many of these proteins also contain a variety of other functional domains.

Figure 2. Schematic suemmary of common features of extracellular matrix adhesion molecules. Linear arrays of structural and functional domains provide a series of specific binding sites.
In general, adhesion molecules frequently have the following properties (1-3, 6, 14-16, 18-25):
• Composed of multiple repeats of protein motifs, such as the immunoglobulin (Ig) motif, the epidermal growth factor (EGF) repeat, or the fibronectin motif.
• Specialized functional domains, including a domain for formation of dimers or higher polymers.
• Moderate affinity, for example, with dissociation constants (Kd) in the range of 10–6–10–7M for fibronectin, and even as weak as 10–4M for leukocyte adhesion molecules that mediate rolling adhesion. In contrast, many well-known protein—protein interactions have affinities with Kd = 10–9–10–11 M.
• High avidity after clustering. Adhesion molecules often form functional clusters or aggregates in the plane of the plasma membrane, which causes them to develop strong total avidity due to the cooperation of the otherwise weak binding of individual molecules.
• Regulation by activation, such as the “inside-out” signaling that increases the affinity of integrins in platelet activation and leukocyte adhesion, or regulation by phosphorylation of the cadherin system.
3. Specific Cell Adhesion Molecules
The broad class of adhesion molecules embedded in the plasma membrane contains several large groups of proteins that share common structural motifs, especially the Ig motif (Fig. 3). Cadherins represent a large superfamily of proteins involved in homophilic cell-to-cell adhesive interactions (14-16). Cadherins bind to cadherins of the same type on other cells via cell interaction sites that can include the recognition sequence His—Ala—Val. Besides the “classic” cadherins, such as E-cadherin and N-cadherin, there are a number of other types of cadherins, as the highly specialized cadherins termed desmocollins and desmogleins found exclusively on desmosomes, which link cells together at these particularly strong attachment sites connected to intermediate filaments such as keratins or vimentin. Cadherins are quite sensitive to depletion of calcium in the surrounding medium, accounting for the ability of calcium chelators such as EDTA to dissociate tissues into component cells. Although desmosomal cadherins are present in epithelia of all ages, cadherins appear to be of particular importance during embryonic development, when they mediate cell—cell adhesion and help define tissues by their propensity to bind primarily to cadherins of the same type, rather than to other cadherins on unrelated cell types (14-16). These specific adhesive interactions result in cell sorting to form tissue regions of closely related cells.
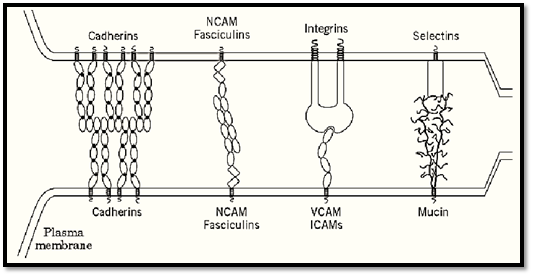
Figure 3. Examples of cell adhesion protein complexes linking plasma membranes of one cell (top) with another (bottom are listed. Adhesion molecules comprising repeating immunoglobulin motifs include cadherins, NCAM, fasciculins, VCA the plasma membrane. Cadherins appear to exist as dimers that form lateral and intercellular connections. Selectins bind carbohydrates in certain mucins. CD2, CD48, and CD58 contain only two Ig repeats. Myelin protein zero (P0) forms hom spaced plasma membranes.
Integrins are major cell-surface receptors for a host of extracellular matrix proteins, as well as “counterreceptors” on other cells (8). There are more than 20 types of integrin subunits. Genetic absence of most integrin subunits leads to disease or death, often in the embryo or near the time of birth. Integrins are heterodimers of one a and one b subunit, each of which provides adhesive specificity. They have a highly distinctive appearance, with a bulbous head domain contributed by each subunit, a binding site that also appears to involve part of each subunit, and two spindly legs (Fig. 3). The legs penetrate through the plasma membrane and usually terminate in rather short cytoplasmic domains. Although short, these cytoplasmic tails can mediate a remarkable range of signaling events. Integrin functions are also often inhibited by depletion of divalent cations.
Although cell adhesion molecules (CAMs) and counterreceptors are structurally related by their use of the Ig motif, and they often even share the -CAM designation, they can differ functionally (Fig. 3). Molecules such as NCAM are homophilic adhesive molecules that bind to the same type of molecule on adhering cell surfaces (22). In contrast, counterreceptors such as the ICAMs and VCAM have specialized peptide recognition sites that are bound specifically by integrins such as LFA-1 (CD11a/CD18 or a1b2) or VLA-4 (a4b1). The functions and sites of expression of these molecules differ widely. For example, molecules such as NCAM are implicated along with cadherins in embryonic developmental events, such as axonal bundling and guidance, whereas the ICAM counterreceptors are present as targets for binding by cells in the blood circulation. Levels of counterreceptors on the cell surface can often also be regulated rapidly in response to cytokines.
A new family of transmembrane proteins termed ADAMs, which are membrane proteins with a disintegrin and a metalloproteinase domain (26), contain a proteinase-like domain, as well as an integrin recognition site in the disintegrin domain. A member of this family is present on sperm and may play a role in adhesive interactions, but the functions of other members of this rapidly growing family remain to be characterized. Syndecans are cell-surface heparan sulfate proteoglycans, with a protein core that crosses the plasma membrane and terminates in a cytoplasmic tail. Syndecans appear to function as “coreceptors,” mediating signaling in association with a primary adhesion molecule (27, 28). For example, syndecans bind to fibronectin at the same time that cell surface integrins bind to a cell-binding domain of fibronectin, and these binding partners cooperate during formation of the specialized adhesion sites known as focal contacts. Syndecans have several other signaling functions as well, and they can to interact with both cytoskeletal proteins and protein kinase C (27, 28).
4. Extracellular Adhesion Molecules
The second major class of adhesive molecules exists extracellularly, and its members are generally targets for cell-surface adhesion receptors such as the integrins. A gallery of such molecules is presented in Figure 4, showing functional binding sites for recognition by cell-surface receptors and domains for binding to a variety of ligands. Many contain one or more heparin-binding domains, which are used to bind to heparan sulfate proteoglycans in the extracellular matrix or in cell-surface syndecan molecules. Many also contain other types of binding domains for extracellular molecules, such as for collagen/gelatin, fibrin, entactin (nidogen), and fibulin (Figs. 2 and 4). Many of these molecules are both large and multimeric, with a specific domain for covalent cross-linking to other subunits. Finally, most of these proteins are either alternatively spliced or encoded by a set of closely related genes, thereby generating a number of isoforms. A number of reviews provide details about these complex proteins.
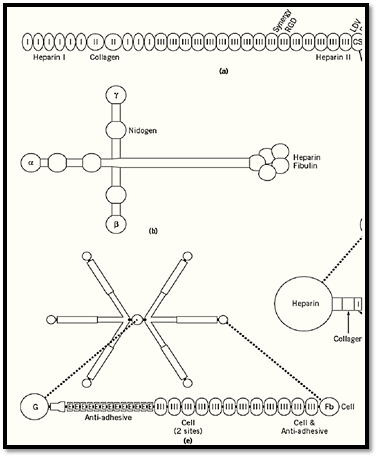
Figure 4. Composite of six examples of cell adhesion molecules of the extracellular matrix: (a) Fibronectin, (b) LamininVitronectin, (d) Thrombospondin, (e) Tenascin, (f) Nidogen (entactin). These molecules vary widely in size and shape, aorganized into functional binding domains. Specific molecular or cell-binding sites are labeled.
5. Peptide Adhesive Recognition Sites
A striking characteristic of a number of cell adhesion molecules is their frequent use of short peptide recognition sites (Refs. 18, 23-25, 29-31 and references cited therein). Short peptides with these sequences can at least partially mimic the binding function of intact molecules to adhesion receptors. Examples include Arg–Gly–Asp (RGD) and Leu–Asp–Val (LDV), which can bind directly to certain integrin receptors. Nevertheless, binding can be 25- to 200-fold more active in larger peptides or proteins, such as in association with a “synergy” site that functions synergistically with the primary adhesive peptide site (30). Some peptide recognition sites appear to be “cryptic” in the intact molecule, and may be active only in small fragments of the protein, such as after proteolysis. Table 1 lists the putative peptide recognition sequences in cell adhesion molecules. It is important to stress that a number of adhesion molecules also function by adhering to other proteins over much broader intermolecular contact areas, via multiple molecular contacts and with no simple peptide motif, in analogy to classic high affinity noncovalent protein–protein interactions (19). It is nevertheless striking that so many cell adhesion molecules use simple, specific, peptide adhesive-recognition motifs.
Table 1. Adhesive Recognition Sequences



Two key sites of membrane adhesion molecules that allow them to function as signal transduction receptors are their ligand-binding domains and their cytoplasmic domains. The ligand-binding domains are of obvious importance in binding to extracellular molecules, but their roles are more intriguing in at least some cases. There appear to be separable functions for ligand occupancy, specifically, for filling the binding site with a ligand or an antibody, as opposed to receptor clustering )which can be induced by a multivalent ligand, such as fibrils of fibronectin or collagen). These two inputs can cooperate in promoting accumulation of specific cytoskeletal proteins, such as a-actinin and actin, which are thought to be crucial for forming strong adhesions (32). Even though they lack intrinsic enzymatic activities, integrins appear to be able to function as signaling receptors and as regulators of actin cytoskeletal organization by recruiting other molecules to their cytoplasmic domains. Integrins can reportedly bind directly to certain cytoplasmic proteins directly, such as to talin, a-actinin, and focal adhesion kinase, perhaps regulated by ligand occupancy on the outside of the cell. Integrin clustering appears to play a central role in forming large intracytoplasmic complexes of over 30 molecules that can serve as signaling centers, such as for MAP kinase activation (Fig. 5; Refs. 9-13, 32).
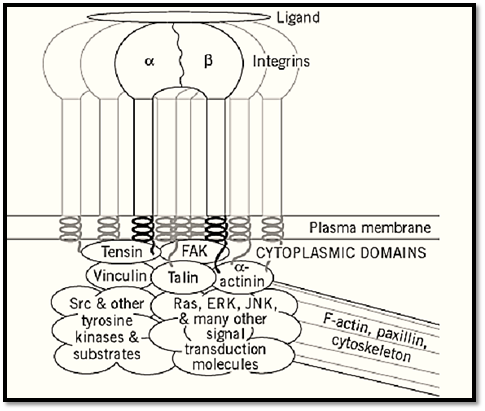
Figure 5. Integrin adhesion and signaling complex formed after cell adhesion molecule interactions. Binding of a ligand and aggregation of adhesion receptors in the plane of the plasma membrane results in the formation of multimolecular complexes that can contain over 30 cytoskeletal and signal transduction molecules, which link to the actin-containing cytoskeleton and mediate intracellular signaling.
6. Major Functions of Cell Adhesion Molecules Beyond Adhesiveness
Cell adhesion molecules and receptors can be intuitively understood as mechanisms for attaching cells to other cells, for permitting cell adherence to extracellular matrix molecules, and for mediating traction during cell migration. It has become clear, however, that they also play critical roles in cellular signaling. In fact, their signaling functions may be of equal biological importance. A current view is that “adhesion” proteins are actually “cell-interaction” proteins that have multiple functions involving the bidirectional transfer of information at the cell surface. In fact, some of these proteins, such as thrombospondin and tenascin, can have antiadhesive activities in certain situations and can modulate a host of cellular functions. Even classic “adhesive” proteins, such as fibronectin, can have a bewildering range of activities, mediated by integrin binding, that range from activating or modulating nearly every known maemmalian signal transduction pathway. These pathways include tyrosine phosphorylation by tyrosine kinases, mitogen-activated protein kinases (MAP kinases,( Ca2+ and H+ fluxes, inositol phosphate pathways, and protein kinase C, which prevent apoptosis and activate specific gene transcription (9-12).
This same theme of complex formation, binding of cytoskeletal proteins such as actin, and accumulation of signaling molecules also appears to occur for cell-to-cell adhesion molecules (14-16, 22). Cadherin cytoplasmic domains interact with catenins, which then link to the actin cytoskeleton. In addition, however, the binding of b-catenin to cadherins can titrate its cytoplasmic levels; b-catenin molecules that are not bound by cadherins can enter the nucleus and regulate gene expression (3). A variety of other signaling pathways exist in cell-cell and cell-matrix adhesion systems. Although much more remains to be learned about precise pathways and overall integration of the many cytoplasmic effects of cell adhesion molecules, it is clear that adhesion molecules are crucial components of the basic regulatory mechanisms of cells and provide dynamic links to the external environment.
References
1. B. Alberts et al., in Molecular Biology of the Cell, 3rd, ed., Garland Publishing, New York , 1994, pp. 949–1009.
2. E.D. Hay, ed., Cell Biology of Extracellular Matrix, Plenum Press, New York 1991.
3. B.M. Gumbiner, Cell 84, 345–357 (1996).
4. S.F. Gilbert, Developmental Biology, Sinauer Associates, Sunderland, Mass., 1994.
5. R.O. Hynes and Q. Zhao, J. Cell Biol. 150, F89–96 2000.
6. G.M. Edelman and J.P. Thiery, The Cell in Contact: Adhesions and Junctions as Morphogenetic Determinants, John Wiley & Sons, New York, 1985.
7. D.L. Benson, L.M. Schnapp, L. Shapiro and G.W. Huntley, Trends Cell Biol. 10, 473–482 (2000) .
8. R.O. Hynes, Cell 69, 11–25 (1992).
9. E.A. Clark and J.S. Brugge., Science 268, 233–239 (1995).
10. M.A. Schwartz, M.D. Schaller, and M.H. Ginsberg, Annu. Rev. Cell Devel. Biol. 11, 549–599 (1995) .
11. A.E. Aplin, A.K. Howe, and R.L. Juliano, Curr. Opin. Cell Biol. 11, 737–744 (1999).
12. F.G. Giancotti and E. Ruoslahti, Science 285, 1028–1032 (1999).
13. K.M. Yamada and B. Geiger, Curr. Opin. Cell Biol. 9, 76–85 (1997).
14. M. Takeichi, Annu. Rev. Biochem. 59, 237–252 (1990).
15. B.M. Gumbiner, J. Cell Biol. 148, 399–404 (2000).
16. U. Tepass et al., Nat. Rev. Mol. Cell Biol. 1, 91–100 (2000).
17. S.K. Sastry and K. Burridge, Exp. Cell Res. 261, 25–36 (2000. (
18. K.M. Yamada, J. Biol. Chem. 266, 12809–12812 (1991).
19. C. Chothia and E.Y. Jones, Annu. Rev. Biochem. 66, 823–862 (1997).
20. R. Gonzalez-Amaro and F. Sanchez-Madrid, Crit. Rev. Iemmunol. 19, 389–429 (1999).
21. M.J. Humphries, Biochem. Soc. Trans. 28, 311–339 (2000).
22.H. Kamiguchi and V. Leemmon, Curr. Opin. Cell Biol. 12, 598–605 (2000. (
23. S. Ayad, The Extracellular Matrix Factsbook, 2nd ed., Academic Press, San Diego 1998.
24. T. Kreis and R. Vale, Guidebook to the Extracellular Matrix, Anchor, and Adhesion Proteins, Oxford University Press, New York 1999.
25. C. Isacke and M. A. Horton, The Adhesion Molecule Factsbook, Academic Press, San Diego 2000 .
26. J. Schlondorff and C.P. Blobel, J. Cell Sci. 112, 3603–3617 (1999).
27. J.R. Couchman and A. Woods, J. Cell Sci. 112, 3415–3420 (1999).
28. A.C. Rapraeger, J. Cell Biol. 149, 995–998 (2000).
29. E. Ruoslahti, Annu. Rev. Cell Devel. Biol. 12, 697–715 (1996).
30. S. Aota, M. Nomizu, and K. M. Yamada, J. Biol. Chem. 269, 24756–24761 (1994).
31. M. Nomizu et al., Arch. Biochem. Biophys. 378, 311–320 (2000).
32. K.M. Yamada and E.H.J. Danen, in J. S. Gutkind, ed., Signaling Networks and Cell Cycle Control, Humana Press, Totowa, NJ, 2000, pp. 1–25.

|
|
|
|
الآثار الجانبية لأدوية تستخدم في علاج "ألزهايمر" تثير الجدل
|
|
|
|
|
|
|
اكتشاف سر نجاة "مخلوقات أبدية" من انفجارات الإشعاع القاتلة
|
|
|
|
|
|
موكب أهالي كربلاء يهدي ممثل المرجعية العليا درعا تثمينا للمساهمات الفاعلة والمساندة لإنجاح الفعاليات التي يقيمها خلال المناسبات الدينية
|
|
|
|
مراحل متقدمة من الإنجاز يشهدها مشروع مركز الشلل الدماغي في بابل
|
|
|
|
الأمين العام للعتبة الحسينية المقدسة: يجب الاهتمام بالباحثين عن العمل ومنحهم الفرص المناسبة عبر الاهتمام بقدراتهم ومؤهلاتهم وإبداعاتهم
|
|
|
|
يمتد على مساحة (500) دونم ويستهدف توليد الطاقة الكهربائية.. العتبة الحسينية تعلن عن الشروع بإنشاء مشروع معمل لتدوير النفايات في كربلاء
|